
报 告 人:彭谦 教授,南开大学
报告地点:北区科技楼三楼报告厅314
报告时间:2021年10月21日13:30-14:30(星期四)
邀 请 人:李吉来 教授
报告摘要:
Demand for higher efficiency, economy, and selectivity in the synthesis of novel molecular scaffolds drives organic chemistry. The development of modular chiral ligands has led to the discovery of several transition metal:ligand complexes that catalyze various reactions with impressive levels of enantioselectivity. However, discovery of the appropriate chiral ligands for a desired transformation remains a formidable task. This is especially true for reactions where detailed mechanistic data are yet to be uncovered. Computational understanding of the mechanism of catalyst-control can lead to improved understanding and guide synthetic effort.
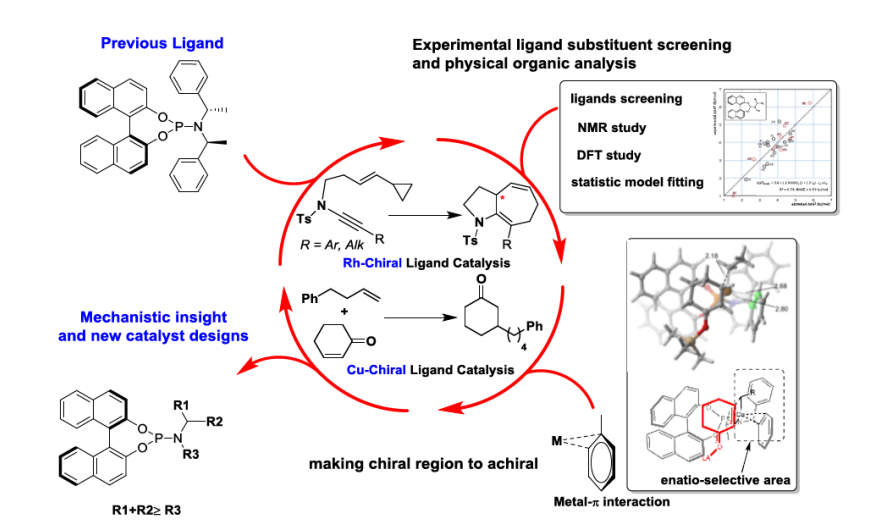
Through computational quantum chemistry we have explored how the structural elements of the phosphoramidite ligand affect catalyst structure and selectivity in Rh-catalysed asymmetry cycloisomerization and Cu-catalysed conjugate additions. Elucidation of the important interactions has been achieved by studying the effects of ligand-structural variation on both the catalyst structure and resulting enantioselectivity, through a combination of experimental and theoretical techniques. These studies illustrate the ability of quantitative structure-selectivity relationships to provide both models for asymmetric induction and catalyst structural hypotheses that may be further probed by experiment and computation. Collectively, such an approach leads to the rational modification/simplification of chiral ligands for more effective catalysts.
报告人简介:
